 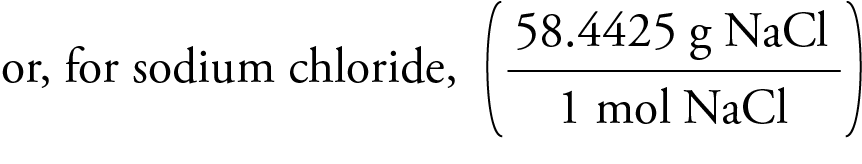
The reaction of carbon dioxide has already been mentioned, but other acidic oxides such as sulfur dioxide (SO 2) also react completely. Related to this is the reaction of sodium hydroxide with acidic oxides. Such acid-base reactions can also be used for titrations, and indeed this is a common way for measuring the concentration of acids. This type of reaction releases heat when a strong acid is used. In general such neutralization reactions are represented by one simple net ionic equation: OH −(aq) + H +(aq) → H 2O The hydroxide ion makes sodium hydroxide a strong base which reacts with acids to form water and the corresponding salts, e.g., with hydrochloric acid, sodium chloride is formed: NaOH( aq) + HCl(aq) → NaCl(aq) + H 2O( l) Sodium hydroxide is completely ionic, containing sodium ions and hydroxide ions. A sodium hydroxide solution will leave a yellow stain on fabric and paper. It is insoluble in ether and other non-polar solvents. It also dissolves in ethanol and methanol, though it exhibits lower solubility in these solvents than does potassium hydroxide. It is very soluble in water with liberation of heat. 
It is deliquescent and also readily absorbs carbon dioxide from the air, so it should be stored in an airtight container. Pure sodium hydroxide is a white solid, available in pellets, flakes, granules, and also 50% saturated solution. Sodium hydroxide is also the most common base used in chemical laboratories, being able to test for quite a number of cations (this is called Qualitative Inorganic Analysis), as well as to provide alkaline mediums for some reactions that need it, such as the Biuret test. Worldwide production in 1998 was around 45 million tonnes. An alkali, caustic soda is widely used in many industries, mostly as a strong chemical base in the manufacture of pulp and paper, textiles, drinking water, and detergents. 
Sodium hydroxide ( Na OH), also known as lye or caustic soda, is a caustic metallic base. Materials in their standard state (at 25 ☌, 100 kPa) Except where noted otherwise, data are given for 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
